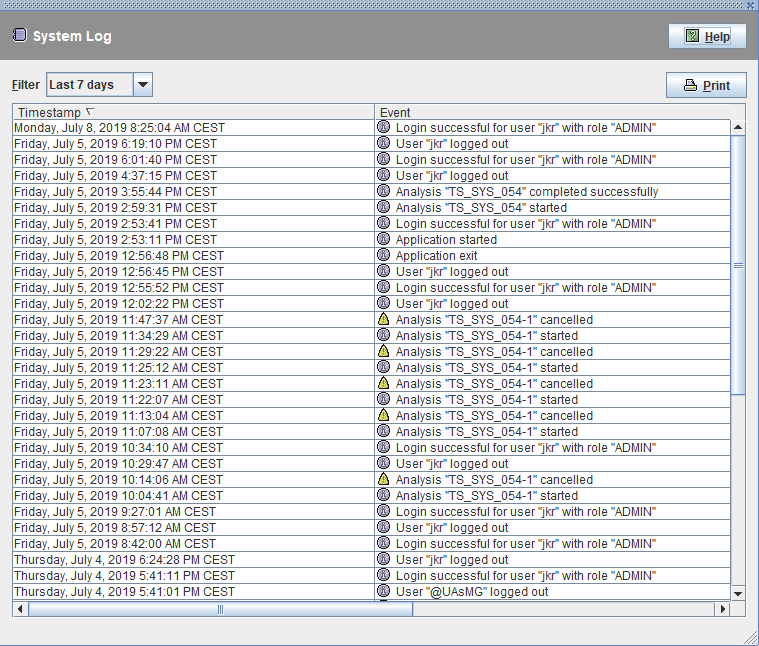
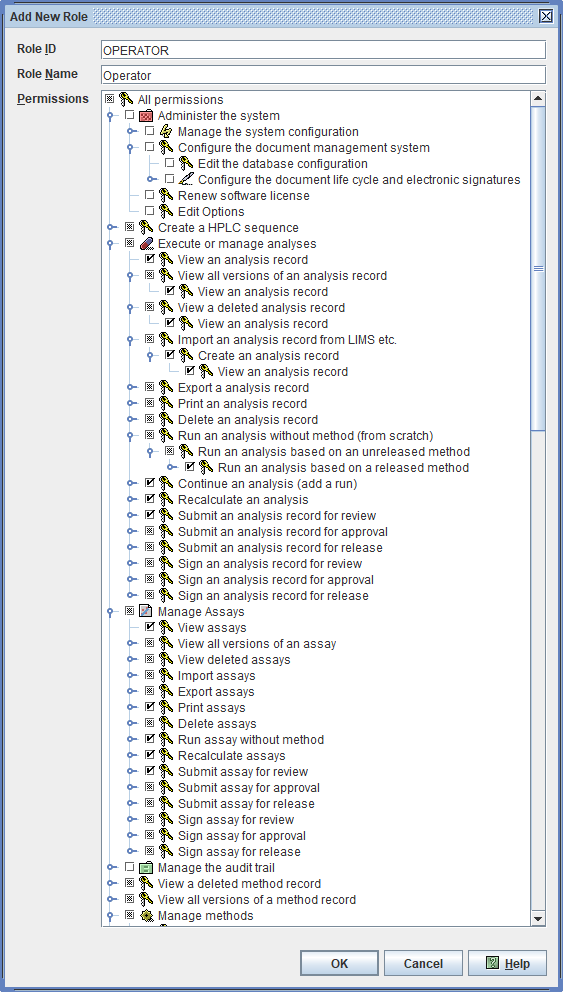
21 CFR Part 11 compliance as well as compliance to the new requirements for Data Integrity
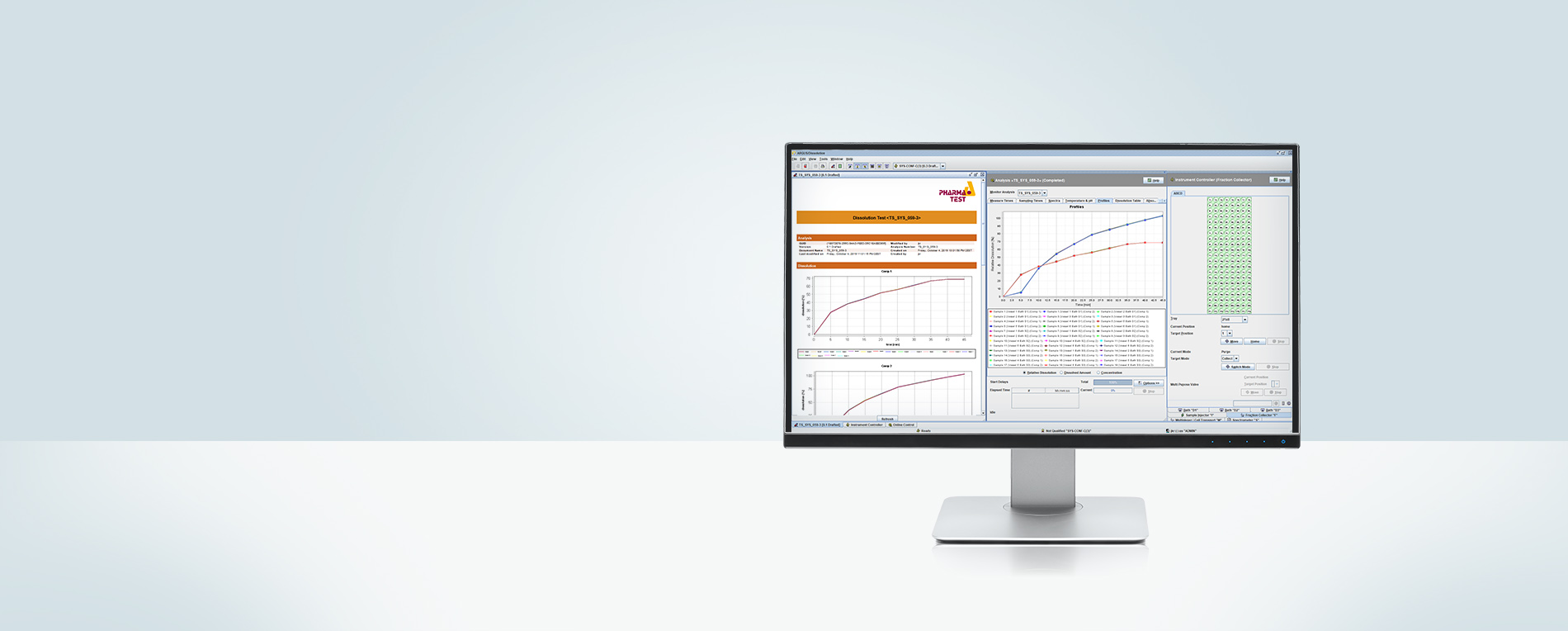
The WinDiss ARGUS Dissolution Software is a scientific dissolution data management software for instrument control, data acquisition, data management, processing and reporting – designed by users for users. WinDiss ARGUS Dissolution is compatible to almost all dissolution systems in the laboratory. Just one software needs to be trained and qualified. Whether you perform dissolution tests occasionally or several times each day WinDiss ARGUS Dissolution will make you more productive in running tests and analyzing results.

The WinDiss ARGUS Dissolution Software installed on the TIDAS ® L 520
WinDiss ARGUS Dissolution simplifies how you collect and report dissolution test results by letting you choose the analysis template that matches the way you work. Whether you are in QC concerned about data integrity, compliance and validation, or need maximum flexibility in R&D, WinDiss ARGUS Dissolution can help your lab perform more efficiently.
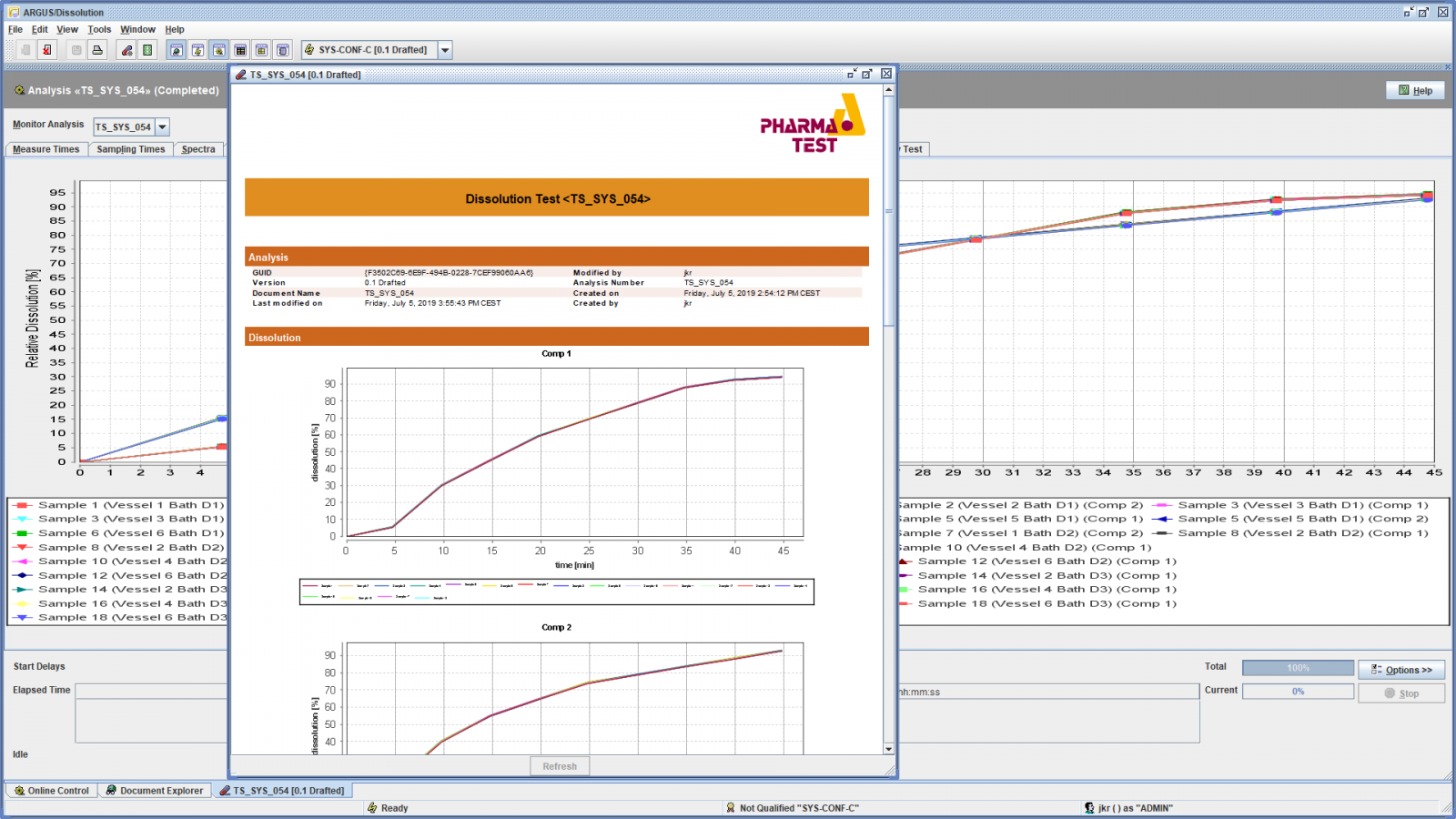
Example Test Report Dissolution Profile
Drivers Currently Available
| Instrument Category |
List of Supported Instruments |
| Drivers for Dissolution Tester |
Pharma Test PTWS 120D/S, PTWS 820, PTWS 620, PTWS 1220, PTWS D620, PTWS 1420, IDS 1000, PT-DT8, PTWS 300, PTWS 310, PTWS 610, PTWS 1210, PTWS D610
Hanson SR8 Plus, Vision Elite & Classic 6, Vision G2 Elite & Classic 6
Sotax AT7/AT7smart, AT Xtend; Varian VK70XX
Distek 2100, 5100, Evolution series
ERWEKA DT800, DT700, DT720
Copley DT600, DT800
|
| Drivers for Spectrophotometers
|
Pharma Test TIDAS L, T70, SA500
Agilent 8453, 8454
Agilent/Varian Cary50
J&M Tidas II (9-channel fiber optic)
AnalytikJena 200/205/210/250
Beckman DU800
Kontron Uvikon XL
Cecil 2000/3000/4000/7000/9000 series
PerkinElmer LAMBDA 2/20/25/35/45
Shimadzu UV1700 PharmaSpec, UV1800
Tec5 MultiSpec
Thermo Evolution 300
Zeiss MCS5XX, MCS6XX(2-channel fiber optic)
|
| Drivers for Pumps |
Pharma Test PT-SP6, PT-SP8
ISMATEC IPC/IPC-N
Hanson AutoPlus MAXIMIZER, Vision G2 AutoPlus
Hamilton Dynamix III
Sotax CY-7-50, CP7
Varian Vk806
Watson Marlow 323Du
Distek Evolution 4300
Solvias Micamix Dual Syringe
|
| Drivers for Dilutor Autosampler & Fraction Collector Driver |
Pharma Test DSR-M, PTFC
Gilson 223 (Agilent G1811A)
Hanson AutoPlus MultiFill, G2 AutoFill
PerkinElmer AS90, AS93plus, S10
Sotax C613/C615
Varian Vk8000
Distek Evolution 4300 Valve Driver
Hamilton MVP
Rheodyne MX Series II Injector Drivers available on demand
Agilent 8452A
Beckman Du600
Distek 2230A
PerkinElmer L265,365,465
|